On Wednesday, City Council signed off on its $160 million plan to change the color of Chicago, replacing its 270,000 high-pressure sodium lights, which give the city its, um, distinctive orange glow, to LED. Right now Chicago is one of the most orange cities in the world; when the project is done, it'll look completely different, on the street and from space.
Chicago's been orange for about 40 years. It started with an experiment on the Dan Ryan in 1969, about the time high-pressure sodium-vapor lights were perfected enough to go into widespread use, and a handful of the blueish mercury-vapor lights were replaced. Three years later the Lawndale People's Planning and Action Conference proposed their light installation on Roosevelt Avenue as a crime-fighting tactic. But it was unclear if the investment would pay off. In 1973, UCLA astronomer Kurt Riegel, concerned about light pollution, correlated rising crime to increased outdoor luminosity in a piece for Science, concluding that "the selling has also been very succesful—most people now believe that outdoor lighting buys them security." (He found that the evidence was mixed.)
Riegel also observed that "high pressure sodium lamps do not account for a very high percentage of outdoor lights in operation presently," but that "municipalities and commercial light users are beginning to install them at a high rate, and the possibility that much of the skylight near urban areas will someday be from this type of lamp should be considered."
He was right. The OPEC oil embargo hit in October 1973, a few months after Riegel's piece was published, and the promise of more light for much less energy was attractive. Riegel plotted out how new technology enabled us to have brighter cities with only a modest increase in energy use.
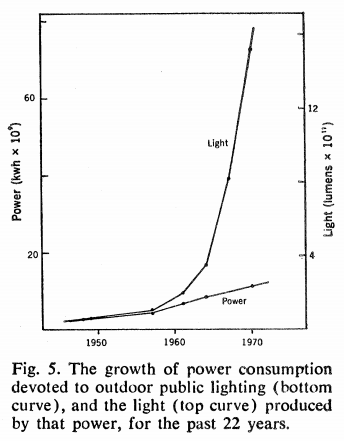
Chicago went fully onboard with high-pressure sodium lights immediately after the OPEC embargo. In November 1973, Mayor Daley requested money for the change. In December the Tribune reported that his plan would make Chicago "the first large U.S. city to have sodium vapor lamps on all residential streets"—plausible, given how new the technology was—replacing the "85,000 harsh metallic-blue mercury vapor streetlights on all residential streets with more cheerful, brighter, gold-colored sodium vapor lamps." In 1976 the city started installing them on its arterial streets.
But as you may have noticed, "cheerful" and "gold-colored" were perhaps an exaggeration. And the city was warned: Trib columnist Jack Mabley spoke with an artist from Vancouver, Ralf Kelman, who had watched his city switch from incandescent lighting to the mercury-vapor lights then common in Chicago. "These [mercury vapor] lights are intense, but they are metallic and harsh. They distort colors and intensity shadows," Kelman said. "They destroy a good mood. A woman goes out in a red dress. She feels great. This light washes her out, turns her dress to mud red. It's ghostly. It produces gloom."
Kelman was barely more enthusiastic about high-pressure sodium lighting. "Well, that will make an orange city," he said. "And when I say orange, I mean orange. It washes everything with orange. It's the lesser evil… better than blue. It's warmer."
It fell on deaf ears in Chicago. Toronto listened, and stuck with incandescent lamps (even as its suburbs went to HPS) until cooler metal halide lamps were rolled out. You can see how metal halide light changes the city in this picture from astronaut Chris Hadfield.

Another skeptic was the Tribune's legendary architecture critic Paul Gapp. "[Sodium vapor lamps] are more than twice as bright as the blue mercury vapor lights which they would replace and produce an artificial daylight effect whose peculiar offensiveness must be visually experienced to be understood. How can it be said?" Gapp wrote in 1974. "One looks at the eerie, ominous quality of sodium vapor illumination and thinks of the bizarre paintings of Hieronymous Bosch, the frightening futurism of Stanley Kubrick's A Clockwork Orange, and other nightmares."
Gapp called up Toronto officials to figure out how and why they banned sodium-vapor lights. It was indeed Kelman, who simply reached out to the city when he found out they were planning on switching to high-pressure sodium, and his arguments caught on, dovetailing with Riegel's observations about crime. "The general, popular reaction in our city was that these orange lights smacked of a police state or a big security system," an assistant to the mayor told Gapp. "The people liked the old [incandescent] lights because they had a pleasant, warm, human quality and did not give the city the look of an armed camp."
"This was an example of the whole citizen access thing we have here," the assistant continued, "which, I guess, is different from what you have in American cities."
Gapp continued railing against the lights, writing that they "have given Chicago the eerie, ominous, after-dark look of a concentration camp" (1976), "grotesque and unnecessary" (in a later 1976 column about how Evanston had shot down the sodium vapor lamps), "quickly took on a symbolism synonymous with danger," "brand the city as one big combat zone," "as a maker of judgments on how our streetscape should look after dark, Daley is strictly a vulgarian" (1976), "the prison yard look" (1978), "sickening" (1980), "absurdly bright and ugly" (1983).
The observations of Kelman, the artist, and Gapp, the architecture critic, have backing in science. Three years ago, Dave Kendricken of No Film School looked at the future of movies in the context of Los Angeles switching from high-pressure sodium to LED, reproducing this spectral power distribution chart from Lamptech:
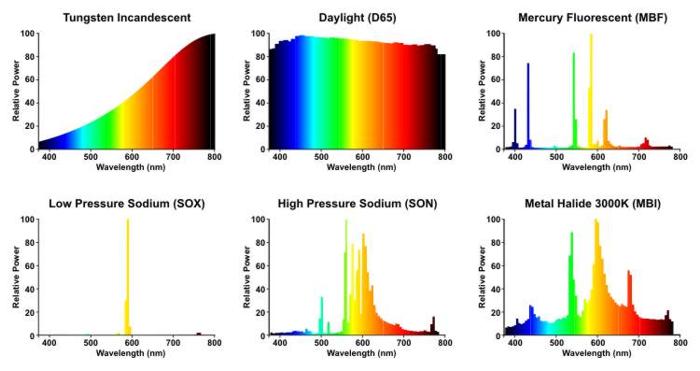
Daylight obviously has the widest spectrum, and it's given a Color Rendering Index of 100—a perfect score. How all other lights render colors are compared to the CRI of daylight.
Mercury vapor (not shown above), the lights that Kelman hated in Vancouver, have a CRI in the 20s. His example of the woman in the red dress may have been a bit dramatic, but he's right about the colors: "While the light source itself appears to be bluish-white, there is a deficiency of long wavelength radiation and most objects appear to have distorted colors. Blue, green, and yellow are emphasized; orange and red appear brownish."
Incandescent lights, which Kelman prevailed on Toronto to keep, reproduce colors very well, and have a CRI near 100, with a wide, warm spectrum, hence the mayoral assistant's description of their "pleasant, warm, human quality." But they're inefficient, so Toronto switched to metal halide when the technology became available. It doesn't offer as wide a spectrum as incandescent, but its CRI is in the 60s to 80s, "suitable for commercial areas," which is why car dealerships use metal halide—color rendering is critical to selling cars.
Typical high-pressure sodium lights have a CRI in the 20s; there are improved versions (CRI of 65 or 70) that you can see in these photos from Stockholm that cover a breadth of streetlights, but they're less efficient. They reproduce colors badly, so a lot of people hate them, but they generate a lot of light very efficiently.
(Low-pressure sodium lights are monochromatic and don't render colors, as you can see in this picture, so their use is uncommon. Hilo, Hawaii, and Flagstaff, Arizona, both near major observatories, make heavy use of low-pressure sodium lights because the fact that they're monochromatic allows astronomers to filter the light, and because it creates less sky glow. As a result, Flagstaff is well regarded among dark-sky proponents, with 60 percent of their streetlights being low-pressure sodium.)
Now LEDs are almost certainly the future of street lights. Looks better, right?

It's a bit more complicated than that. High-pressure sodium lamps produce very little blue light. LEDs—or at least some of the ones that cities started to install as street lights—produce a lot. And even though it arguably looks a lot better, once they started going in, people started to think that maybe a wider spectrum wasn't what we wanted. Think about how people use light at different times of day:
An incandescent bulb has a color temperature of 2400K, which means it contains far less blue and far more yellow and red wavelengths. Before electric light, we burned wood and candles at night; this artificial light has a [color temperature] of about 1800K, quite yellow/red and almost no blue. What we have now is very different.
High-pressure sodium is around 2200K, in between fire and incandescent light. The first round of LED streetlights had color temperatures between 4000K and 5000K—cool and blue, like the fluorescent lights lots of us work under, and closer to daylight—because lower temperatures are less energy efficient.
LEDs started becoming popular as researchers were looking into the effects of artificial lighting on the body. There's a reason the lights we use during the daytime are cool, but they may not be suited for nighttime:
Blue wavelengths—which are beneficial during daylight hours because they boost attention, reaction times, and mood—seem to be the most disruptive at night. And the proliferation of electronics with screens, as well as energy-efficient lighting, is increasing our exposure to blue wavelengths, especially after sundown.
[snip]
While light of any kind can suppress the secretion of melatonin, blue light at night does so more powerfully. Harvard researchers and their colleagues conducted an experiment comparing the effects of 6.5 hours of exposure to blue light to exposure to green light of comparable brightness. The blue light suppressed melatonin for about twice as long as the green light and shifted circadian rhythms by twice as much (3 hours vs. 1.5 hours).
The American Medical Association noted this research, and called for cities to install LED streetlights of 3000K or less, though there's been pushback from the Municipal Solid-State Streetlighting Consortium, which argues that the lower light output of LEDs makes up for its higher blue content.
Chicago followed the AMA's guidelines and is getting 3000K LEDs. It's close to the 2700K lights the city of Davis, California, got after its citizens revolted against the "prison lighting" of their new 4000K LEDs, or Phoenix, which switched to warmer lights under public pressure, not the only city to field complaints about cool-temperature lights. Again, Ralf Kelman, the artist, called it: "And when I say orange, I mean orange. It washes everything with orange. It's the lesser evil… better than blue. It's warmer."
What does the difference look like? On the left is a high-pressure sodium light. On the right is a 4000K light. In the middle is approximately what we're getting.

It's a compromise. Incandescent lights are beloved because they're warm and their color-rendering is excellent, so they feel natural and have a good warmth for nighttime lighting—but, they're extremely inefficient. High-pressure sodium lights are warm, but their color rendering is terrible ("the ugliest light known to the cinematographer"). LED streetlights have much better color rendering, but it's unnatural to have the night lit like the day—in ways we can perceive, and perhaps in ways we can't. Chicago's 3000K LEDs are an attempt to have it both ways: familiar enough in rendering colors to not look like "frightening futurism," warm enough to be appropriate for the night. Somewhere, Paul Gapp is nodding.



